Innovation based on experience leads to novel and safe medical devices.
emka medical was founded by Dr. Raymond Glocker in 2011 to manifest his vision of a truely stable and continuous invasive blood pressure measurement for monitoring of heart failure. Having already worked since 1990 on similiar projects he knew from experience what was lacking in the available technology. For this reason emka medical developed the first closed fluid filled catheter.

Our mission
We want to give patients with cardiopulmonary diseases the best possible hemodynamic monitoring during their treatment. We believe true optimization of medication or device based therapy can only be achieved by getting direct and continuous feedback of the therapy on core parameters like pressure inside the heart.
We believe data driven therapy is the only way to achieve best possible treatment of the patients. At the same time we want to keep quality of life high by reducing restrictions like tubings and cables to enable as much mobility as possible. That is why we have developed CorLog.

1990
Pressure catheter

2005
Reusable pressure transducers and domes

2008
Implantable intermittent pressure catheter
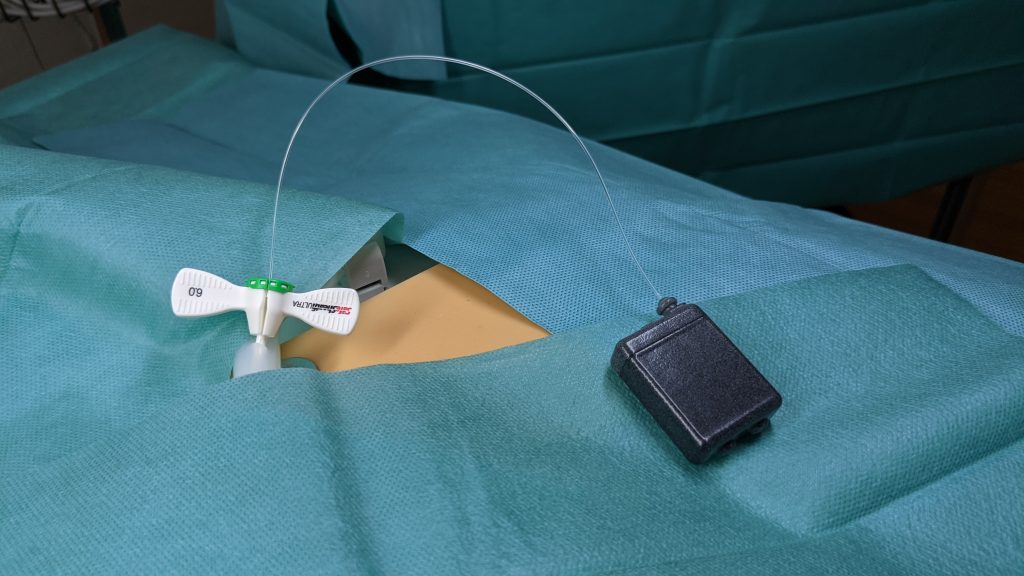
2018
Continuous stable pressure probe
Our Management

CEO
Uwe Damerau
Chief Executive Officer
CFP Lawyer, graduated from the European Business School. Independent financial advisor for 30 years.

Dr.rer.nat.
Raymond Glocker
Chief Operating Officer
Being a solid state and bio physicist with a PhD degree, he has over 30 years of experience in the medical device industry.

Prof. Dr. med.
Hans-Bernd Hopf
Chief Medical Officer
Professor of Anaesthesia, Emergency and Intensive Care Medicine, Chief Emeritus of the Asklepios Klinik Langen

CQO
Roger Staab
Chief Quality Officer
Seasoned regulatory and quality management veteran with 15 years of experience in the medical device business.
The history of our company
2011
- CGS Sensors GmbH was founded as Spin-Off from MHM Harzbecher Medizintechnik
2014
- First software as medical device CE approval for Cor/log View
2015
- Development of first in world closed fluid filled pressure sensitive catheter
2016
- Seal of Excellence by the European Horizon 2020 R&D program
- Funding from Bavarian Ministry of Economy and Technology(BayMED)
- Start of project CorLog Probe 1P as joint venture between CGS Sensors and emka TECHNOLOGIES
- Successful preclinical use of pressure catheters for 3 months
2017
- CGS Sensors is renamed to emka medical
2018
- First passed audit according to ISO 13485:2016
- Finalized CorLog System
2019
- First in man use of CorLog
2020
- First use of CorLog in COVID-19 Pneumonia/ARDS patients
2021
- Successful 180 days implanted in preclinical tests
- CorLog App gets CE approval
2022
- First use of CorLog in pulmonary hypertension patients
- First passed EU-MDR Audit
Our Team












Advisory Board (GmbH Beirat)
Jean-Gérard Napoléoni started Emka Technologies (MKT) in 1992 where he was CEO until 2021 when he sold the company. MKT has a worldwide footprint, developing, manufacturing and selling technical equipment and infrastructure to measure cardiopulmonary function in preclinical laboratories, in particular through implanted devices.
Walter Heindl is a financial expert with more than 25 years of experience. He hands-on acts as angel investor, board member, and consultant, primarily focuses on Life Sciences. He held leadership positions in several leading investment banks, including Country Head of Germany & Austria, Vice Chairman of Asset Management, Global Head of Wholesale Strategy, and regional CAO for Asia ex-Japan.
Uwe Damerau, CFP Lawyer, graduated from the European Business School. Independent financial advisor for 30 years.
Attorney Philipp Georgi has been working in the field of corporate law in Berlin for many years. In addition to the preparation of notarial contracts (establishment, structuring, financing rounds), he advises startups from the development of their products to the exit. Attorney Georgi has a large network at his disposal.
